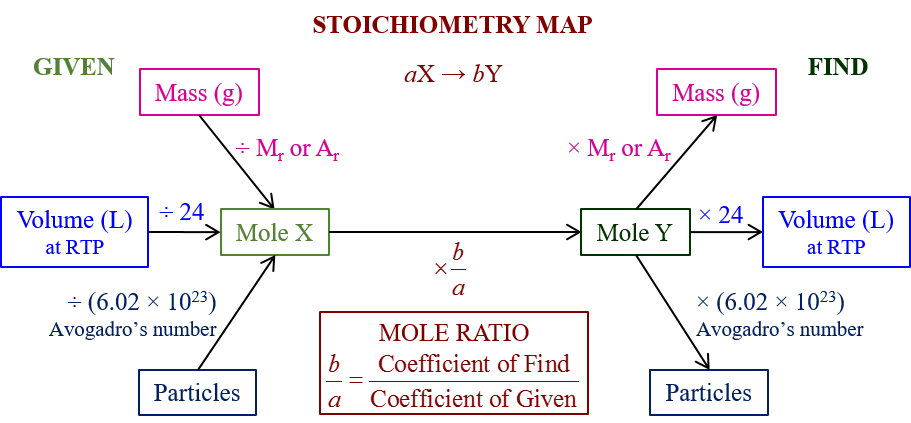
These equations can be used to calculate the energy per mole the fuel releases.Thermochemical equations can be used to compare fuels.Using the equation- 1 mole of methane releases 890KJ of energy.įor 1250 moles, Energy = 1250/1 x890 = 1112500KJ (using ratios) Thus to you need to work the given equation and given mass together in a stoichiometric fashion to determine:ġ.How much mole of the fuel you actually have.Ģ.The actual energy released for this number of mole.Ĭalculate the energy released, in MJ, when 20.0 kg of methane undergoes complete combustion.ĬH4(g) + 2O2 (g) -> CO2(g) + 2H2O (l) ΔH= -890KJ/mol.The coefficients of the reactants in a thermochemical equation indicate the amounts (moles) of each substance that reacts to give the specified enthalpy change.Volume ratio is same as molar ratio because of constant pressure and temperature. 
Step 2: From equation find ratio of V(unknown) to V(known). What volume of oxygen is needed to react with 10 mL of hydrogen sulfide?ĢH2S (g) + 3O2 (g) -> 2H2O (g) + 2S02 (g) Hydrogen sulfide may be oxidised to sulfur dioxide and water. If the gases are at the same pressure and temperature, their molar ratios are equal to their volume ratios.įinding volume of gas when conditions of pressure and temperature are constant.Chemical reactions where all reactants and products are gases.Step 4: Answer Question – Find Volume of propane. Step 3: From equation find ratio of n(unknown) to n(known). Step 1: Write a balanced equation and identify known and unknown.Ĭ3H8 (g) + 5O2 (g) ->3CO2 (g) + 4H20 (g) 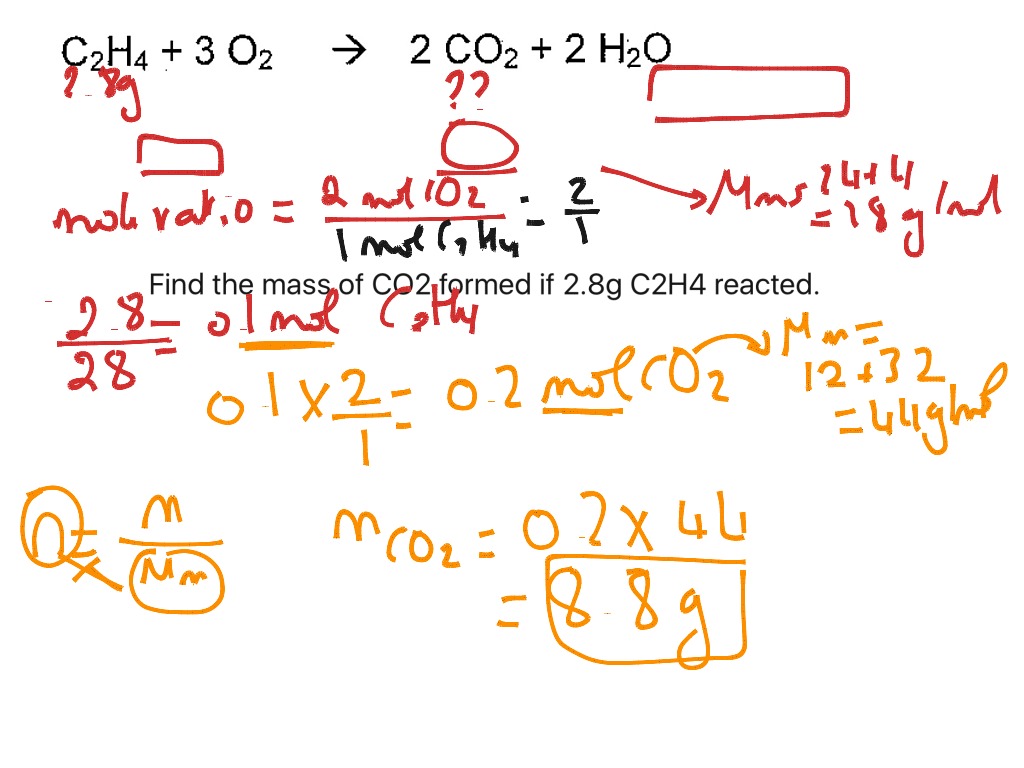
22.0 g of CO2 is collected and weighed.įind the volume of propane at 200oC and 101 325 Pa. In a gas barbeque, propane was burnt in oxygen to form carbon dioxide and water vapour.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
